
High-dose flu shot may lower risk of Alzheimer's

Do gallstones always need treatment?

Healthier plant-based diet tied to lower risk of dementia

Is MRI contrast dye safe?

Are those body aches a sign of gallstones?

Staying active throughout middle age may lower women's risk of dying early

Hantavirus explained: What to know after the cruise ship outbreak

Stay strong at any age with 4 basic exercises

Don't count on daily aspirin to prevent colon cancer

After ablation, exercise may lower atrial fibrillation recurrence
Staying Healthy
Achieving and maintaining good health means adopting healthy lifestyle habits, from consuming nutritious foods and getting regular physical activity to prioritizing sleep and reducing stress.
Dietary patterns that are most closely linked to a longer, healthier life tend to focus on unprocessed or minimally processed foods like fruits, vegetables, legumes, whole grains, and nuts as well as fish and healthy fats.
Physical activity can mean going for long walks, riding your bike, gardening, playing pickleball, or regularly participating in any activity you enjoy. Exercise lowers your risk for serious health problems, strengthens and tones your muscles, helps preserve independence, and boosts your mood.
Restorative sleep is similarly vital to your health and wellbeing, as is managing stress. Regular checkups, screening tests, and other preventive health measures are also important.
Over time, these efforts will pay dividends. You’ll be rewarded with more energy, improved mobility, less pain, a brighter outlook, and enhanced physical and mental health.
Related Articles

Digestive Health
Are those body aches a sign of gallstones?
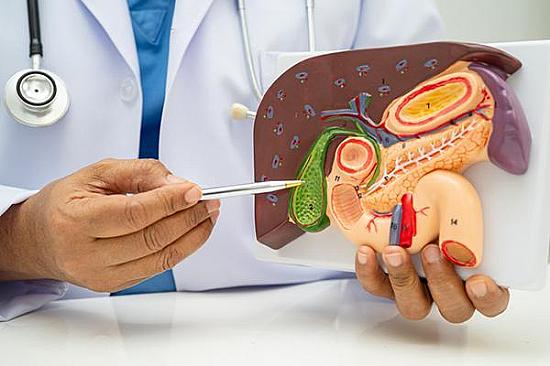
Digestive Health
Do gallstones always need treatment?

Heart Health
Treating high blood pressure may help lower risk of cognitive decline

Mindfulness and meditation
Video: Recharge your mind with mindfulness

Staying Healthy
Tackling the top stressors for dementia caregivers

Staying Healthy
Are you healthy enough to age in place?

Exercise and Fitness
How Steady Are You? A Balance & Fall-Prevention Quiz
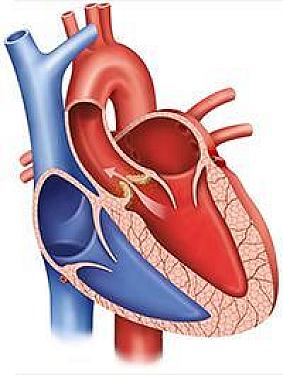
Heart Health
Heart-healthy habits over time linked to a lower risk of aortic stenosis

Exercise and Fitness
The best stretching routines for men

Digestive Health
The dos and don'ts of managing diverticular disease

Mind & Mood
Exercise can boost your memory and thinking skills

Heart Health
What's the best blood pressure target for older adults?
Free Healthbeat Signup
Get the latest in health news delivered to your inbox!
Sign Up

