New view of heart disease in women
A revolution in thinking about coronary blood vessels could change the way women's heart problems are diagnosed and treated.
Heart disease is the leading cause of death in women." There, we've said it again. Even though it's always been the case, we still need to remind ourselves that we're far more likely to succumb to heart disease than to breast cancer. Nine times as likely, in fact.
The statistics are alarming, but researchers are beginning to make sense of them. Findings from the federally funded Women's Ischemia Syndrome Evaluation (WISE) study — a landmark investigation into ischemic heart disease in women — are helping us to understand that heart disease, like cancer, is not one, but several disorders. Discoveries from the WISE study suggest that many women have a form of heart disease called coronary microvascular dysfunction that isn't detected by standard diagnostic procedures and thus goes unrecognized and untreated. The research, led by Dr. Noel Bairey Merz at Cedars-Sinai Medical Center in Los Angeles, is laying the groundwork for better ways to evaluate and treat women with heart disease.
Overturning conventional wisdom
The WISE study is upending a decades-old paradigm in which heart disease was thought to be gender-neutral. Heart attacks are less likely to occur in premenopausal women than in men the same age, a benefit long attributed to natural estrogen. But doctors also assumed that when women developed heart disease, they had the same condition as men: atherosclerotic plaque restricting blood flow in the coronary arteries that nourish the heart muscle.
As a result, the procedures used to diagnose and treat heart disease were based on research conducted only in men. In this unisex approach, people with symptoms such as chest pains first undergo noninvasive stress tests to determine whether the heart is getting enough blood as oxygen demands increase. If these tests indicate reduced blood flow to the heart, the next step is coronary angiography — an x-ray that shows where the vessel is blocked and the size of the obstruction. Once pinpointed, the blockage is flattened during coronary angioplasty or bypassed with a vessel graft.
In the 1970s, the National Institutes of Health launched the Coronary Artery Surgery Study to evaluate this standard protocol. It was the first major heart study to include women, who made up about a quarter of the 25,000 participants. The findings were baffling: A woman with a positive stress test was 4.5 times more likely than a man with a positive stress test to have an angiogram showing no blockages. The researchers took this to mean that stress testing was unreliable in women. They also concluded that women had a much lower incidence of coronary artery disease than men. The cause of the women's chest pain was left unexplored.
The next few decades saw major advances in the diagnosis and treatment of coronary artery disease. The cardiovascular death rate declined steadily — but only in men. Women had angioplasties and bypasses, but they didn't do as well as their male counterparts. More of them suffered heart attacks or congestive heart failure after treatment. Even women whose coronary arteries were clear continued to develop and die from ischemic heart disease (IHD). By 2000, heart disease was claiming 60,000 more women than men every year.
|
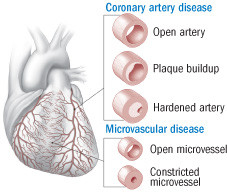
Causes of ischemic heart disease
Ischemic heart disease (IHD) is reduced blood flow to the heart. According to the classic model of the disease, trouble begins when plaque builds up inside a coronary artery, which eventually thickens and hardens, obstructing blood flow. The newer view of IHD suggests that the tiny vessels feeding the heart can also become constricted and reduce blood flow. |
Medicine WISEs up
In 1996, the NIH decided that these gender disparities couldn't be ignored any longer. Not only were more women dying of heart disease, but female heart patients required longer hospital stays and incurred higher medical bills than males.
The WISE study was designed to answer three questions: How does IHD develop in women without arterial blockages? Would some other cardiac imaging method provide better clues to IHD in such women? What role do estrogen and other female hormones play?
The researchers enrolled nearly 1,000 women who had chronic chest pain severe enough to warrant coronary angiography. The women were interviewed about their chest pain, quality of life, and history of depression. They underwent laboratory tests for cardiac risk factors and traditional diagnostic procedures, including stress testing and coronary angiography. But they also received some newer tests, including intravascular ultrasound, which evaluates the motion and structure of the blood vessel wall. Women with blocked coronary arteries had angioplasty or bypass. All the participants were monitored for at least a year for ongoing chest pain, heart attack, or stroke.
As expected, many symptomatic women with positive stress tests had negative angiograms. But many of these women also had abnormal results on vascular function tests. Their coronary arteries and smaller vessels feeding the heart muscle didn't dilate properly and thus couldn't accommodate increased blood flow — a condition known as vascular dysfunction. Vascular dysfunction occurred both in women with clear arteries and in those with obvious coronary artery narrowing. Moreover, it was not a problem of the large coronary arteries alone, but involved stiffening of the network of smaller vessels that also nourish the heart. This condition has been dubbed coronary microvascular dysfunction, or microvessel disease, and is sometimes referred to as Syndrome X.
A "Eureka!" moment
To some cardiologists, the discovery of widespread microvessel disease came as an epiphany. It helped explain why so many women with IHD were misdiagnosed and undertreated: The standard protocol identified only coronary artery obstructions. Microvessel disease could also help explain why so few women have the classic crushing chest pain that signals coronary artery disease. Instead, they feel diffuse discomfort, exhaustion, or shortness of breath under stress or even during daily routines — symptoms that are nonspecific and less dramatic than those that herald a blockage caused by a blood clot. As vessels lose their resilience, blood flow is reduced, and the heart muscle, deprived of oxygen, gradually dies, resulting in congestive heart failure.
The WISE data may also explain why women who undergo angioplasty and bypass surgery don't fare as well as men. These women may have both coronary artery disease and unrecognized microvessel disease. In such cases, opening the arteries isn't sufficient.
|
Coronary artery disease and coronary microvascular dysfunction compared |
||
|
Coronary artery disease |
Coronary microvascular dysfunction |
|
|
Common symptoms |
Crushing chest pain often radiating to the left arm, cold sweat, nausea |
Diffuse discomfort, exhaustion, depression, shortness of breath |
|
Pathology |
Isolated plaque lesions extending into the vessel lumen |
Constricted vessels |
|
Diagnostic procedures |
Stress test, coronary angiography |
Stress test, functional vascular imaging |
|
Treatment |
Angioplasty, bypass surgery, statins |
Anti-hypertensives, anti-inflammatories, statins |
Probing the pathology
How and why does coronary microvascular syndrome develop? Inflammation is a prime suspect. For one thing, inflammation is implicated in atherosclerosis. It's also at the heart of inflammatory disorders, such as autoimmune diseases, which are far more common in women than in men.
The WISE team found that three proteins associated with inflammation — C-reactive protein (CRP), interleukin-6 (IL-6), and serum amyloid A (SAA) — were particularly important. Compared to women with low levels of these proteins, women with the highest levels were at much greater risk for a coronary event or death within five years — even though they were only slightly more likely to have coronary artery blockages. This led to the idea that inflammatory markers could help assess microvessel disease in women.
Autopsy studies support this approach. They show that women who die of heart attacks often have a pathology different from that of their male counterparts. In men, plaque tends to form in isolated accumulations that jut into the lumen (the open passage through which the blood flows) or burrow into the artery wall. In women, plaque is more likely to be deposited uniformly around the inside of the vessel — the possible handiwork of chronic inflammation.
When inflammatory substances course chronically through the bloodstream, they strip away cells that line the blood vessels, creating an ideal bed for cholesterol-laden plaque. The plaque gave these women's vessels stiff interior walls that tended not to rupture under stress (a common characteristic of heart attacks in men) but, rather, to erode. Efforts to repair the erosion perpetuate the inflammatory cycle and spur the formation of tiny blood clots, a possible trigger for heart attacks resulting from microvessel disease.
Beyond inflammation
Inflammation isn't the only cause of coronary microvascular dysfunction. The WISE researchers looked at the classic cardiovascular risk factors and found that all the usual suspects — genes, high LDL cholesterol, low HDL cholesterol, high triglycerides, elevated blood sugar, hypertension, sedentary lifestyle, and obesity — were implicated. They uncovered some other risk factors as well, including these:
Premenopausal high blood pressure. Hypertension, particularly in younger women, is a major risk factor for IHD. It damages the cells lining the coronary vessels, which attracts immune cells that trigger and feed inflammation.
Anemia. The WISE study determined that cardiovascular outcomes are worse for anemic women than for women with normal hemoglobin levels. Anemia leaves fewer red blood cells to supply the heart muscle with oxygen.
Polycystic ovarian syndrome (PCOS). Women with PCOS have many components of the metabolic syndrome (a risk factor for heart disease) along with erratic levels of circulating estrogen. Estrogen may dampen inflammation, so when too little is available, the risk of heart disease may rise, especially in premenopausal women.
|
Selected resource "Insights from the NHLBI-Sponsored Women's Ischemia Syndrome Evaluation (WISE) study," Parts I and II, Journal of the American College of Cardiology (Feb. 7, 2006), vol. 47, No. 3, Suppl. 1, pp. S4–S20 (Part I) and S21–S29 (Part II). |
What's the wisdom for us?
The WISE investigators are developing a new protocol for screening women for heart disease, but it may be years before this process is completed. Meanwhile, here are some things you can do right now to reduce your risk for microvessel disease.
Live heart-healthfully. Take the same steps you would to prevent coronary artery disease: Get regular exercise; maintain a healthy weight; don't smoke; and keep cholesterol levels in balance, glucose in check, and blood pressure under control.
Pay attention to symptoms. Certain seemingly unrelated symptoms — fatigue, depression, shortness of breath — could be preludes to a serious cardiac event. Don't be shy about seeking medical help for these symptoms.
Talk to your doctor. If you're at high risk for or have symptoms of heart disease, such as unexplained fatigue or shortness of breath, discuss the WISE findings with your clinician. She or he may want to reevaluate your care, especially if you've had a "false-positive" stress test (that is, the stress test indicated a problem but further testing was negative).
Reassess your risk. Even if you're not at risk for coronary artery disease, your chance of an early cardiac event might be elevated by microvessel disease. The odds are higher for African American women (who tend to suffer heart attacks at an earlier age than whites) and premenopausal women with inflammatory disorders, autoimmune diseases, or low estrogen levels. If you're in one of these groups, do all you can to reduce your lifestyle risk factors.
Disclaimer:
As a service to our readers, Harvard Health Publishing provides access to our library of archived content. Please note the date of last review or update on all articles.
No content on this site, regardless of date, should ever be used as a substitute for direct medical advice from your doctor or other qualified clinician.