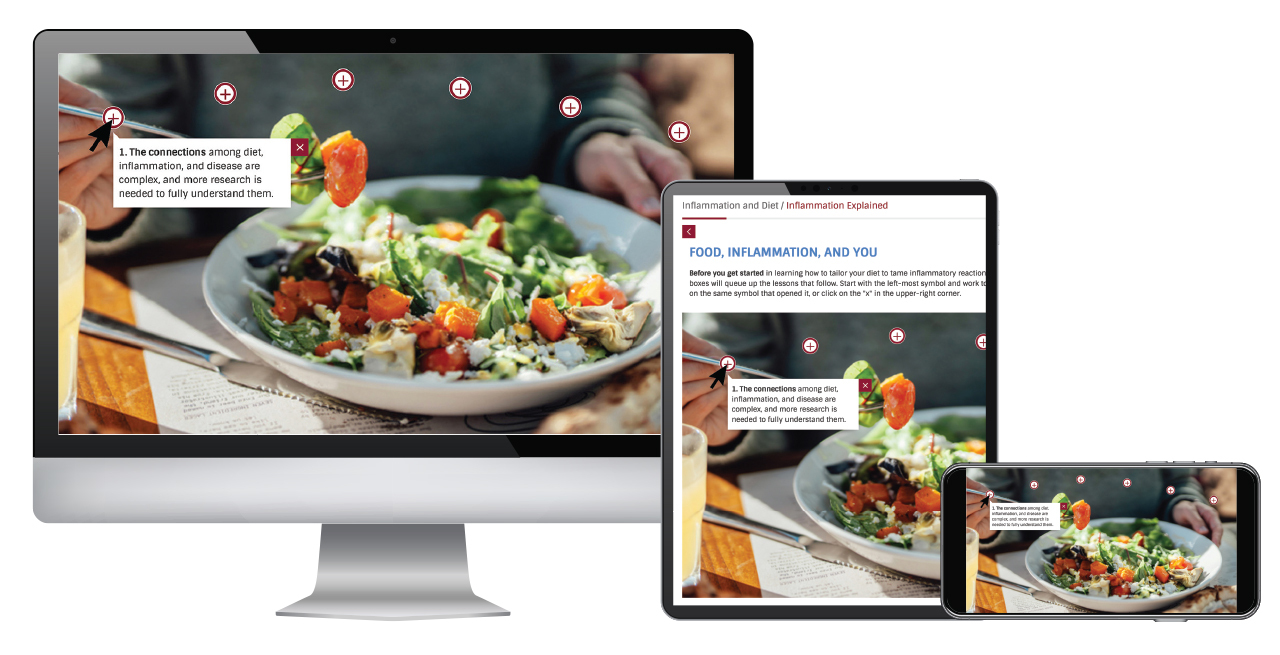
Coronavirus Resource Center

COVID-19 (coronavirus disease 2019) is a respiratory illness caused by the SARS-CoV-2 virus. It is very contagious, and spreads quickly.
Most people with COVID-19 have mild respiratory symptoms that feel much like a cold or flu. But it can be much more serious for older adults, people with underlying medical conditions, and those who haven’t been vaccinated against COVID-19. Some people may go on to suffer from post-COVID conditions, known as “long COVID.” Mounting evidence suggests that COVID vaccines protect against long COVID.
Vaccines against COVID-19 are the best defense against the virus, as they protect against serious disease, hospitalization, and death.
Even if you have been vaccinated, you will want to take steps to reduce your chances of infection. That may mean temporarily wearing masks indoors or avoiding large gatherings if COVID-19 levels are high in your area. The good news is these steps will also reduce your risk of developing other respiratory viruses, like colds or flus, too.
We know a lot more about COVD-19 than we did in 2020, and yet we're still learning. We will continue to provide important updates.
FAQs
No results matched your search.
Terms to know
antibodies: proteins made by the immune system to fight infections. If the antibodies later encounter the same infection, they help prevent illness by recognizing the microbe and preventing it from entering cells.
antigen: a substance displayed on the surface of a microbe that stimulates the body to produce an immune response.
antigen test: a diagnostic test that detects specific proteins on the surface of the virus.
diagnostic test: indicates whether you are currently infected with COVID-19.
epidemic: a disease outbreak in a community or region.
false negative: a test result that mistakenly indicates you are not infected when you are.
false positive: a test result that mistakenly indicates you are infected when you are not.
immunity: partial or complete protection from a specific infection because a person has either had that infection previously or has been vaccinated against it.
incubation period: the period of time between exposure to an infection and when symptoms begin.
infectious respiratory particles: droplets containing respiratory pathogens that can spread from person to person when a sick person coughs, sneezes, or talks.
isolation: the separation of people with a contagious disease from people who are not sick.
long COVID: the constellation of symptoms experienced by COVID long-haulers. These symptoms may include brain fog, shortness of breath, chills, body ache, headache, joint pain, chest pain, cough, and lingering loss of taste or smell.
long-haulers: people who have not fully recovered from COVID-19 weeks or even months after first experiencing symptoms.
mutation: A change to a virus’s genetic material that occurs when the virus is replicating. The change is passed on to future generations of the virus.
mRNA: short for messenger ribonucleic acid, mRNA is genetic material that contains instructions for making proteins.
mRNA vaccines: mRNA vaccines for COVID-19 contain synthetic mRNA. Inside the body, the mRNA enters human cells and instructs them to produce the "spike" protein found on the surface of the COVID-19 virus. The body recognizes the spike protein as an invader, and produces antibodies against it. If the antibodies later encounter the actual virus, they are ready to recognize and destroy it before it causes illness.
pandemic: a disease outbreak affecting large populations or a whole region, country, or continent
polymerase chain reaction (PCR) test: a diagnostic test that detects the presence of the virus's genetic material.
SARS-CoV-2: short for severe acute respiratory syndrome coronavirus 2, SARS-CoV-2 is the official name for the virus responsible for COVID-19.
spike protein: a protein on the surface of the SARS-CoV-2 virus that binds to and allows the virus to enter human cells.
variant: A virus containing one or more mutations that make it different from a version of the virus that has been circulating.
virus: a virus is the smallest of infectious microbes, smaller than bacteria or fungi. A virus consists of a small piece of genetic material (DNA or RNA) surrounded by a protein shell. Viruses cannot survive without a living cell in which to reproduce. Once a virus enters a living cell (the host cell) and takes over a cell's inner workings, the cell cannot carry out its normal life-sustaining tasks. The host cell becomes a virus manufacturing plant, making viral parts that then reassemble into whole viruses and go on to infect other cells. Eventually, the host cell dies.
Image: Naeblys/Getty Images